 This is not consistent with the dative bonding in coordination compounds where it is assumed that one partner donates an electron pair and the other partner accepts it. The valence bond concept in its original form assumes that each bonding partner contributes one electron to the covalent bond. The concept works very well to explain the shapes of molecules of main group elements. These orbitals can either be atomic orbitals, or hybridized atomic orbitals. 
The basic idea is to overlap half-filled valence orbitals to form covalent bonds in which the two electrons are shared between the bonding partners (Fig. Figure 7.1.1 Electron sharing and valence bond theory 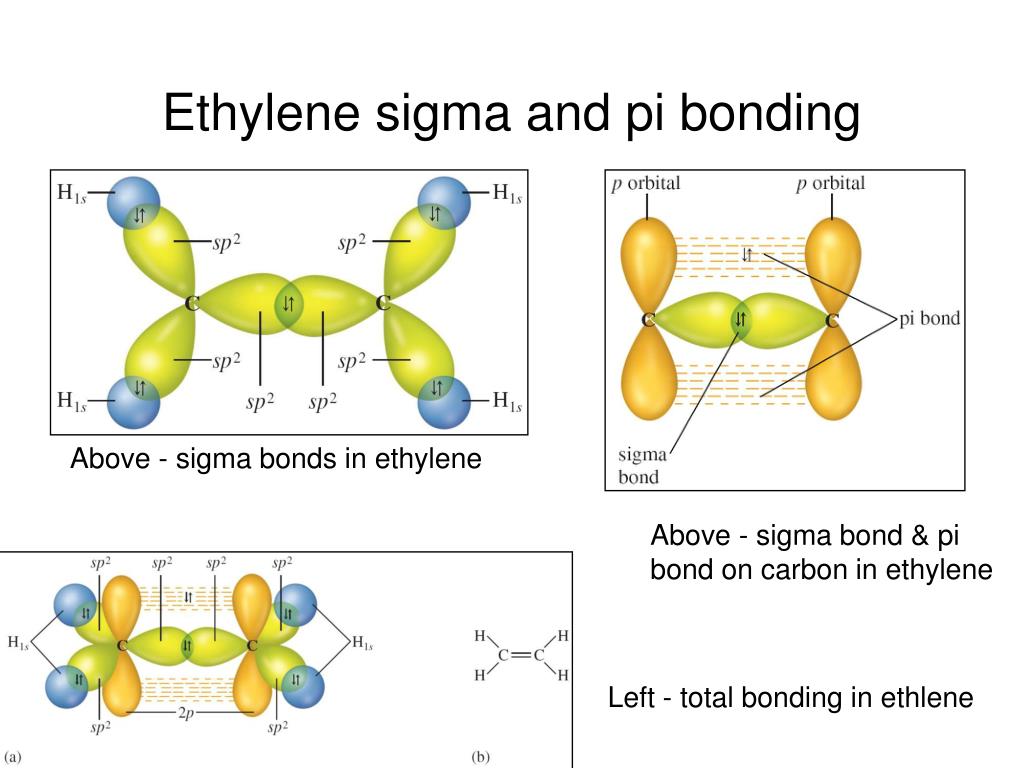
The valence bond concept was introduced by Linus Pauling in 1931 to explain covalent bonding in molecules of main group elements. The first one is the valence bond theory. There are essentially three bonding concepts that are used to describe the bonding in coordination compounds. Optical properties of compounds are linked to bonding because they are related to electronic states. It should further be able to explain the stability and reactivity of complexes, as well as the optical properties of complexes. A paramagnetic molecule is attracted by an external magnetic field. A diamagnetic molecule is repelled by an external magnetic field. It is paramagnetic when there are unpaired electrons. Remember, a molecule is diamagnetic when it has no unpaired electrons. In addition, it should be able to explain the magnetism of molecules, in particular dia- and paramagnetism. What does this mean for a bonding theory? What would a good bonding theory for coordination compounds be able to do? It should certainly be able to explain and predict the number of bonds and the shape of a molecule. The more the theory can explain and predict, and the fewer the necessary assumptions, the better the theory. In addition, it should be able to predict experimental observations. The answer is, that it should be able to make many correct explanations for experimental observations based on a few, sensible, assumptions. Let us first think about, what a good theory should be able to do in general. This chapter is devoted to bonding theories for coordination compounds. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
